Refining Methods
Contents
Refining Methods
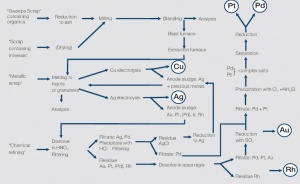
The precious metal containing products and materials submitted into the refining process are generally called precious metal scrap Figure 1. They are usually classified into three categories which differ by the refining processes employed and by the cost-to-value ratio.
Refining of Meltable Scrap
Precious metal scrap that can be re-melted in a technically economical way includes:
- Scrap from electrical and electronic processes such as precious metal
plates or strips, wires, stamping skeletons, stamping scrap, etc.
- Metal scrap from the optical industry, i.e. precious metal containing
frames for glasses
- Process scrap from the jewelry and watch manufacturing as well as
obsolete jewelry
- Metal scrap from the dental industry
The refining process starts with melting of a homogeneous alloy in granulate or ingot form. Using different analytical methods the exact precious metal content is determined. Once verified the actual refining and separation of the precious metal components is performed using a sequence of electrochemical and chemical processes.
The widely used electrolytic refining of silver performed by the “Moebius” – process has great economical importance. Plates of continuously cast highsilver-content material are dissolved as the anode in a nitric acid electrolyte solution. Through the influence of an electrical field the silver ions move to a steel cathode where they are reduced to silver metal and deposited as pure silver in the form of crystalline dendrite particles. The process is adjusted in a way that allows the non-precious components to stay in solution or be collected as sludge in bags that surround the silver anodes. The silver reclaimed in this way has a purity of 99.99%. After relatively simple cleaning drying process silver is usually re-melted into ingots or granulate and returned into the manufacturing process, e.g. for contact materials.
Low Mixed Content (Sweeps) Refining
“Low Grade Scrap” or “Sweeps” (also called “Dross”) is precious metal containing scrap which is mixed with organic or mineral components, such as precious metal containing polishing and grinding residues, sludge from spent gold and silver electrolytes, paint or resin containing residues, ion exchange compounds, filter papers, precious metal catalysts on substrate carriers, and also floor sweeps from jewelry shops, dental laboratories, and other industrial manufacturing operations. Such residues cannot be re-melted and therefore require special processing steps. First the scrap material is incinerated in a special ash-producing furnace. In this process metal and mineral residues remain in the ashes while organic compounds are vaporized. After grinding and screening the metal containing ash particles are separated and processed further as re-meltable scrap. The precious metal containing ash is homogenized through a special process and used for representative sampling to determine the exact precious metal content.
Chemical Refining
Special refining scrap, i.e. used precious metal electrolytes, salts, gold plated printed circuit boards, IC scrap, metallic scrap with high melting components such as Ag/W, or composite materials like Ag/Ni, Ag/MeOx and others, precious metal pastes or paste coated substrates, cannot be refined economically in the processes described above.
Figure 2 Processes for the refining of precious metal scrap
The term chemical refining is used for all refining processes that employ the classic separation methods – dissolving, precipitation, filtration, washing. As opposed to the other methods, the chemical refining process rarely allows the collection of a representative sample. The precious metal amounts contained in the supplied scrap can usually only be determined at the completion of processing. Only for electrolytes and rinse waters is it possible to obtain the expected amount of recovered precious metals through an upfront chemical analysis.