Difference between revisions of "Physical Properties of the Most Important Metals"
| Line 228: | Line 228: | ||
<div id="text-reference"><sub>1</sub> at 20°C</div> | <div id="text-reference"><sub>1</sub> at 20°C</div> | ||
| − | <xr id=" | + | <xr id="tab:Atomic Properties of the Most Important Metals"/> |
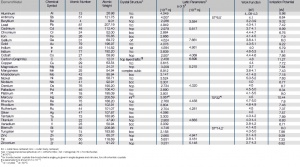
'''Tab. Atomic Properties of the Most Important Metals''' | '''Tab. Atomic Properties of the Most Important Metals''' | ||
<br /> | <br /> | ||
| − | <xr id=" | + | <xr id="tab:Thermal Properties of the Most Important Metals"/> |
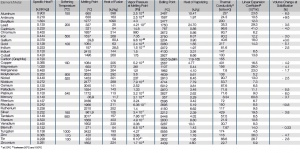
'''Tab. Thermal Properties of the Most Important Metals''' | '''Tab. Thermal Properties of the Most Important Metals''' | ||
<br /> | <br /> | ||
| − | <xr id=" | + | <xr id="tab:Electrical Properties of the Most Important Metals"/> |
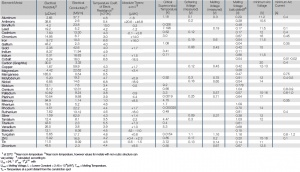
'''Tab. Electrical Properties of the Most Important Metals''' | '''Tab. Electrical Properties of the Most Important Metals''' | ||
<div class="multiple-images"> | <div class="multiple-images"> | ||
| − | < | + | <figtable id="tab:Atomic Properties of the Most Important Metals"> |
[[File:Atomic-Properties-of-the-Most-Important-Metals.jpg|left|thumb|Atomic Properties of the Most Important Metals]] | [[File:Atomic-Properties-of-the-Most-Important-Metals.jpg|left|thumb|Atomic Properties of the Most Important Metals]] | ||
| − | </ | + | </figtable> |
| − | < | + | <figtable id="tab:Thermal Properties of the Most Important Metals"> |
[[File:Thermal-Properties-of-the-Most-Important-Metals.jpg|left|thumb|Thermal Properties of the Most Important Metals]] | [[File:Thermal-Properties-of-the-Most-Important-Metals.jpg|left|thumb|Thermal Properties of the Most Important Metals]] | ||
| − | </ | + | </figtable> |
| − | < | + | <figtable id="tab:Electrical Properties of the Most Important Metals"> |
[[File:Electrical-Properties-of-the-Most-Important-Metals.jpg|left|thumb|Electrical Properties of the Most Important Metals]] | [[File:Electrical-Properties-of-the-Most-Important-Metals.jpg|left|thumb|Electrical Properties of the Most Important Metals]] | ||
| − | </ | + | </figtable> |
</div> | </div> | ||
<div class="clear"></div> | <div class="clear"></div> | ||
Revision as of 16:56, 30 April 2014
The following tables list the physical properties of the most technically significant pure metals as well as carbon. The values given may vary considerably depending on the degree of purity and sometimes they are also difficult to determine. In compiling the data from the available literature we selected those that are currently the most probable. Some properties are anisotropic and vary with the crystalline structure of the metal. In those cases, whenever possible, we listed the value applicable to the poly-crystalline stage.
Tab. Mechanical Properties of the Most Important Metals
| Element/Metal | Density 1
[g/cm³] |
Modulus of
Elasticity 1[GPa] |
Shear Modulus
[GPa] |
Transvers ContractionCoeffic. |
|---|---|---|---|---|
| Aluminum | 2.70 | 65 | 27 | 0.34 |
| Antimony | 6.62 | 56 | 20.4 | 0.28 |
| Beryllium | 1.85 | 298 | 150 | 0.12 |
| Lead | 11.36 | 14.5 | 6 | 0.44 |
| Cadmium | 8.65 | 57.5 | 29 | 0.30 |
| Chromium | 7.19 | 160 | 0.25 | |
| Iron | 7.89 | 208 | 83 | 0.28 |
| Gallium | 5.91 | 9.6 | 0.46 | |
| Gold | 19.32 | 79 | 28 | 0.42 |
| Indium | 7.31 | 11 | 0.45 | |
| Iridium | 22.65 | 538 | 214 | 0.26 |
| Cobalt | 8.85 | 216 | 0.31 | |
| Carbon (Graphite) | 2.1-2.3 | 5 | ||
| Copper | 8.95 | 115 | 48 | 0.34 |
| Magnesium | 1.74 | 46 | 18 | 0.28 |
| Manganese | 7.43 | 165 | 77 | 0.24 |
| Molybdenum | 10.21 | 347 | 122 | 0.30 |
| Nickel | 8.90 | 216 | 83 | 0.31 |
| Niobium | 8.57 | 113 | 39 | 0.38 |
| Osmium | 22.61 | 570 | 220 | 0.25 |
| Palladium | 12.02 | 124 | 51 | 0.39 |
| Platinum | 21.45 | 173 | 67 | 0.39 |
| Mercury | 13.55 | |||
| Rhenium | 21.04 | 480 | 215 | 0.26 |
| Rhodium | 12.41 | 386 | 153 | 0.26 |
| Ruthenium | 12.45 | 485 | 172 | 0.29 |
| Silver | 10.49 | 82 | 27 | 0.37 |
| Tantalum | 16.60 | 188 | 70 | 0.35 |
| Titanium | 4.51 | 120 | 43 | 0.34 |
| Vanadium | 6.10 | 136 | 52 | 0.36 |
| Bismuth | 9.80 | 33 | 13 | 0.33 |
| Tungsten | 19.32 | 360 | 158 | 0.30 |
| Zinc | 7.13 | 96 | 36 | 0.29 |
| Tin | 7.30 | 47 | 18 | 0.33 |
| Zirconium | 6.49 | 98 | 36 | 0.33 |
Table 1
Tab. Atomic Properties of the Most Important Metals
Table 2
Tab. Thermal Properties of the Most Important Metals
Table 3
Tab. Electrical Properties of the Most Important Metals
References
Metals Handbook, Desk Edition: Chicago, IL, American Society of Metal, 1985
Landolt-Börnstein: Zahlenwerte und Funktionen. Springer-Verlag, Berlin-Göttingen-Heidelberg, 1959
Handbook of Chemistry and Physics, 70th Edition: CRC Press., Inc. Boca Raton, Florida, 1989 - 1990
Fluck, E.; Heumann, K., G.: Periodensystem der Elemente. Weinheim: VCH-Verlagsgesellschaft, 1986
Kieffer, R.; Jangg, G.; Ettmayer, P.: Sondermetalle. Springer- Verlag, Wien-New York, 1963
Hering, E.; Schulz, W.: Physik für Ingenieure (Periodensystem der Elemente). Düsseldorf: VDI-Verlag, 1988
Degussa AG (Hrsg.): Edelmetall-Taschenbuch. Hüthig-Verlag, Heidelberg, 1995
Slade, P.; G. (editor): Electrical Contacts Principles and Applications. Marcel Dekker, Inc., New York-Basel, 1999
Gerritsen, A.; N.: Metallic Conductivity in: Flügge, S.: Handbuch der Physik, Bd. 19, Springer-Verlag, Berlin-Göttingen-Heidelberg, 1956
Köster, W.; Franz, H.: Poisson,s Ratio for Metals and Alloys. Metallurg. Reviews 6 (1961)
Nesmeyanow, A., N.: Vapor Pressure of the Chemical Elements: Elsevier, Amsterdam-London-New York, 1963
Wyckoff, R., W., G.: Crystal Structures. Vol 1,New York, 1963